 And don’t forget to put the unit g/mol to your final calculated molar mass. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in IrON: Molar Mass (g/mol) Ir (Iridium) 1 × 192.217 192.217. 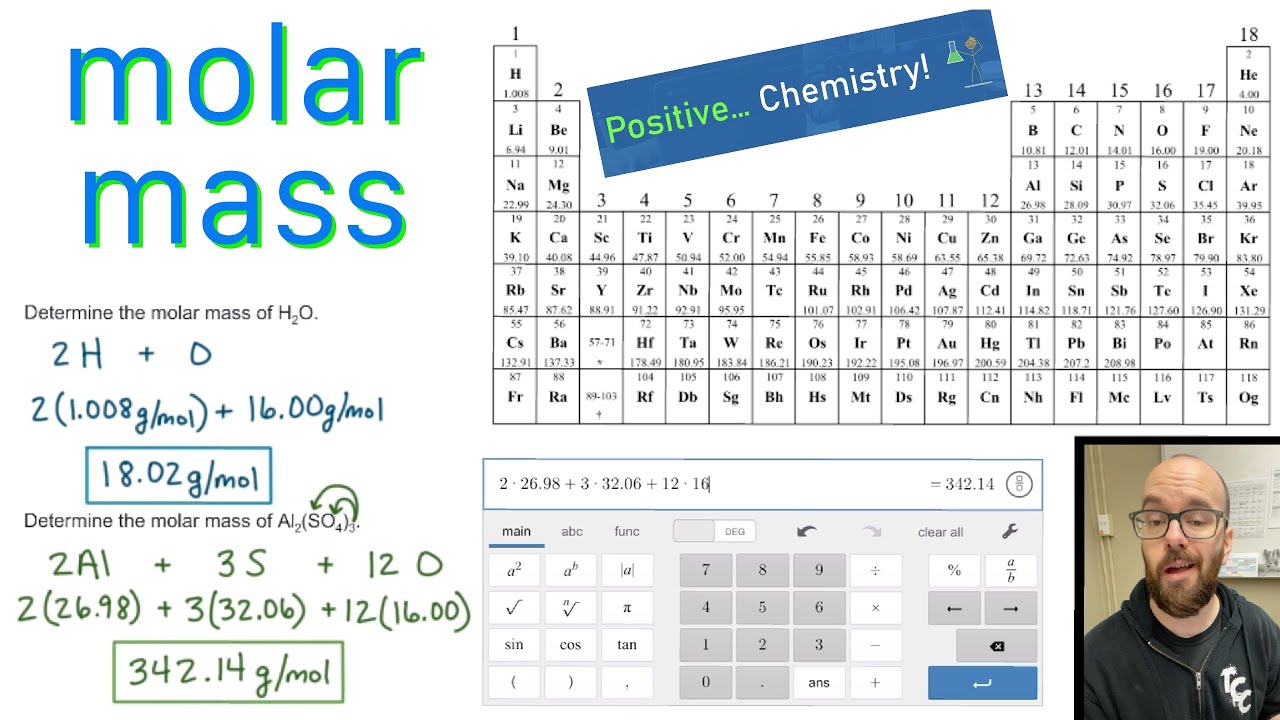
First solve the brackets, then multiplications and at last do the final addition. Always follow the calculation order to avoid any mistakes in calculation.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole.See original paper for the range of these elements from different sources Isotope-abundance variations and atomic weights of selected elements: 2016 (IUPAC Technical Report), Pure Appl. I hope you have understood the short and simple calculation for finding the molar mass of Sucrose. List of Elements with Range of Atomic Weights. At the very bottom is the name of the element (e.g., hydrogen). Below is the relative atomic mass, as calculated for the isotopes found naturally on Earth. In the middle is the letter symbol for the element (e.g., H). At the upper left is the atomic number, or number of protons. Because the masses of all other atoms are calculated relative to the 12 C standard, 12 C is the only atom listed in Table 2.3.2 whose exact atomic mass is equal to the mass number. Please notify us of any errors or corrections in the above data. The arbitrary standard that has been established for describing atomic mass is the atomic mass unit (amu or u), defined as one-twelfth of the mass of one atom of 12 C. Hence the Molar mass of Sucrose (C12H22O11) is 342.397 g/mol. Image showing the 'anatomy' of a periodic table entry. Authors not responsible for errors, omissions or inaccuracy of the above data. So, Molar mass of Sucrose (C12H22O11) = Molar mass of 12 Carbon (C) atoms + Molar mass of 22 Hydrogen (H) atoms + Molar mass of 11 Oxygen (O) atoms. Since protons and neutrons contribute the. 
In terms of weight, one unified atomic mass unit is equivalent to one-twelfth of the mass of a Carbon-12 atom at rest. Determining Molecular Weights Using Wet Chemistry. :max_bytes(150000):strip_icc()/PeriodicTablecolor-58b5d9105f9b586046df7d8a.jpg)
Unified atomic mass units are commonly used to quantify atomic mass. His great work, published in 1895, also gave important insight into the atomic theory of matter. 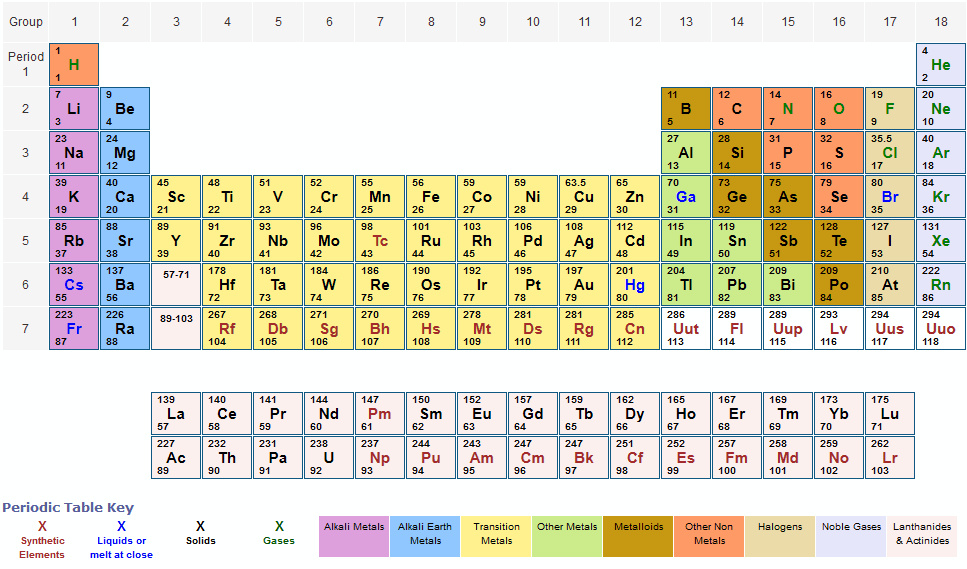
Visualize trends, 3D orbitals, isotopes, and mix compounds. You can see that in Sucrose (C12H22O11), there are 12 Carbon atoms, 22 Hydrogen atoms and 11 Oxygen atoms. The atomic mass of an element is a measure of the average mass of its atoms, expressed in Atomic Mass Units (AMU). Interactive periodic table showing names, electrons, and oxidation states. \]Ĭarbon is predominantly 12C, so its average atomic mass should be close to 12 amu, which is in agreement with this calculation.Now, to calculate the molar mass of Sucrose, you just have to add the molar mass of all the individual atoms that are present in Sucrose. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
